Friday, August 20, 2010
Use of Dispersants in the Gulf Proves to Have Little Benefit
Dr. David Valentine - one of the world's leading experts on oil-eating bacteria - told me today:
We have found no Alcanivorax borkumensis in the deepwater plumes.
That may not seem like a very interesting or controversial sentence.
But it is actually a jaw-dropping statement, showing that the use of Corexit in the Gulf has failed by any measure.
To understand why, let's quickly run through the science of the oil spill.
Why Was Corexit Used in the Gulf?
While many - including me - have accused BP of dumping millions of barrels of Corexit in the Gulf in order to hide the oil, some scientists argued that Corexit would make it easier for oil-eating microbes to break down the oil.
As Scientific American pointed out in May:
The last (and only) defense against the ongoing Deepwater Horizon oil spill in the Gulf of Mexico is tiny—billions of hydrocarbon-chewing microbes, such as Alcanivorax borkumensis. In fact, the primary motive for using ... chemical dispersants on the oil slick both above and below the surface of the sea is to break the oil into smaller droplets that bacteria can more easily consume.
In fact, there is no clear science showing that dispersants help microbes break down oil. As Chemical and Engineering News noted in June:
Behind those teams’ assumption lie murky data. The authors of the 2005 [National Research Council] dispersant report described the results of three decades of research into dispersants’ effects on biodegradation as “mixed” with studies showing evidence for “enhancement, inhibition, and no effect.”
Nevertheless, tremendous quantities of Corexit have been sprayed some 5,000 feet underwater at the wellhead.
Does Corexit Interfere with the Microbes?
Dr. Valentine - a biogeochemist at the University of California, Santa Barbara - received funding from the National Science Foundation to characterize the microbial response to the Gulf oil spill. The National Science Foundation has funded Dr. Valentine's research into how the oil-eating microbes are dealing with the spill, and whether or not Corexit is interfering with the microbes' ability to break down the oil.
As I wrote last month:
Corexit might be killing the oil-eating bacteria which would otherwise break down the oil. University of Georgia scientist Samantha Joye notes that scientists have no idea how the large quanties of dispersant will effect the Gulf's microbial communities (for more information, watch part 1, part 2, part 3, part 4 and part 5 of Dr. Joye's July 13th press conference).
Moreover, as MSNBC notes, oil-eating bacteria are less active in deepwater, where much of the oil sinks after treatment with dispersants:
Some note that little is known about the deepwater ecosystem — or how the oil and dispersants will react under extremely high water pressure, very low temperatures, limited oxygen and virtually no light.
***
The conditions at the bottom of the Gulf also could affect the bacteria that help break down the oil near the surface, as they are less active in cold temperatures than in the warm surface waters, and they may be less abundant in the deep.
“We know that the surface material has been degrading,” says Ralph J. Portier, professor of environmental studies at LSU. “But what about the microbial population at depth?”
As Scientific American points out:
[But] colder, deeper waters inhibit microbial growth. "Metabolism slows by about a factor of two or three for every 10 degree[s] Celsius you drop in temperature," notes biogeochemist David Valentine of the University of California, Santa Barbara, who just received funding from the National Science Foundation to characterize the microbial response to the ongoing oil spill. "The deeper stuff, that's going to happen very slowly because the temperature is so low."Mother Jones provides additional details:
***
At the same time, the addition of ... dispersants deep beneath the surface is having uncertain effects; it may even end up killing the microbes it is meant to help thanks to the fact that Corexit 9527A contains the solvent 2-butoxyethanol, which is a known human carcinogen and toxic to animals and other life.
David Valentine ... warns the stuff may be riskier than just its toxicity. Corexit may undermine the microbes that naturally eat oil.
Some of the most potent oil-eaters—Alcanivorax borkumensis —are relatively rare organisms that have evolved to eat hydrocarbons from naturally occurring oil seeps. Valentine tells Eli Kintisch at Science Insider that after spills, Alcanivorax tend to be the dominant microbes found near the oil and that they secrete their own surfactant molecules to break up the oil before consuming the hydrocarbons. Other microbes don't make surfactants but devour oil already broken into small enough globs—including those broken down by Alcanivorax.
What we don't know is how the surfactants in Corexit and its ilk might affect the ability of Alcanivorax and other surfactant-makers to eat oil. Could Corexit exclude Alcanivorax from binding to the oil? Could it affect the way microbes makes their own surfactants? Could Corexit render natural surfactants less effective?
The National Science Foundation has awarded Valentine a grant to study the problem.
Alcanivorax borkumensis is the main microbe breaking down oil in the Gulf. For the real science nerds, here are two illustrations of how breaks down oil (click either image for full graphic):
No Microbes in Deepwater PlumesWhen I spoke with Dr. Valentine today, I asked him whether he has reached any conclusion as to whether Corexit interferes with the ability of Alcanivorax borkumensis to break down the crude.
He replied that he doesn't yet have any findings on that issue.
However, he said, "We have found no Alcanivorax borkumensis in the deepwater plumes".
I asked him whether that was because of the cold temperatures deep under the surface of the ocean, and he replied "we don't know".
I had been worried that the microbes would break down the crude in the underwater oil plumes more slowly than normal ... but Valentine said there are no microbes at all down in the plumes.
The mongoose was introduced to Hawaii in order to control rats (the rats were eating the sugar cane used to make rum, and so were a real nuisance).
It didn't work out very well - mongeese are daylight-loving creatures while rats are nocturnal . So the mongeese didn't control the rats, and instead ran amok and trashed many native Hawaiian species.
The scientists who assumed that Corexit would help the microbes break down the oil from the Gulf spill haven't fared much better.
Remember, the US Minerals Management Service and a consortium of oil companies - including BP - found that as little as 2% of the oil which spills from deepwater wells ever makes it to the surface of the ocean. As Alexander Higgins noted in June:
As the University of California Santa Barbara notes, microbes like the lighter portions of the oil:[A] study called Project “Deep Spill” ... analyzed a wide range of controlled releases at different depths below the sea surface of different types of oil found all over world to help better understand the flow of hydrocarbons released from a deepwater blowout.
One of the studies, called DeepBlow, released 10,000 barrels of oil per day at a depth of 800 meters which is less than half of the depth of the Deepwater Horizon blowout.
The basic findings of that study has been recreated by scientists from the University of North Carolina.
In their research the scientists simulated of the formation of the underwater oil plumes that are created during deepwater blowouts.
Watch The University of North Caroline Simulation Shows How Oil Released Underwater Forms Plumes
While the University of North Carolina simulation gives you a basic understanding of how deepwater blowouts create oil plumes it does not fully account for all the findings of Project “Deep Spill”.
In particular the final report of Project “Deep Spill” found:
1. Only 2% of the oil released in a deepwater blowout may actually make it to the surface. That’s as little as 2% naturally without the use of dispersants. Add dispersants into the equation and it could be less then one percent of oil that makes it to the surface.
***
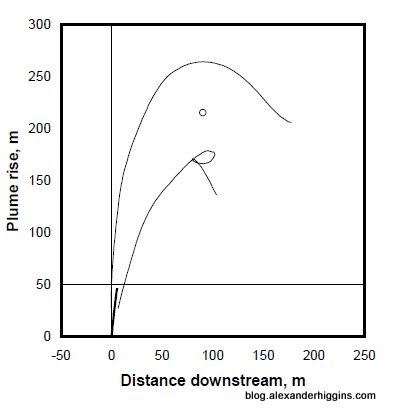
3. The buoyant parts of the oil released in a deepwater blowout split from the main plume within the first 200 meters of release. Those buoyant parts, which represent only a small portion of the total amount of oil, turn into small droplets that float to the surface.
Here is a graph from the study showing this process.
Deepwater oil release – Buoyancy particle separation graphHere is an image that captures the separation process
Deepwater oil release – Buoyancy particle separation simulation4. Within the first 100 to 200 meters from the source of the release the the majority of the oil loses its buoyancy and stops rising. This majority of the oil remains submerged in an underwater plume that is then carried away by subsurface currents.
The microbes prefer the lighter compounds of oil, the gasoline part of the black goo ...."There always seems to be a residue," Valentine said. "They (bacteria) hit a wall. There seems to be stages in which they eat. There's the easy stuff ––the steak. And then they work their way to the vegetables, and then garnish, and then they stop eating after awhile. Just depends on how hungry they are and what's fed to them."
Corexit: A Failed Science Experiment
So let's recap.
The overwhelming majority of oil from deepwater spills stays beneath the surface in plumes. See this and this.
Oil-eating microbes like the lighter portions of oil.Writing in the journal Science, the scientists confirmed that oil from the well had been caught below the surface of the Gulf in pools of microscopic oil drops and petroleum-based trace chemicals, which were degrading more slowly than many had expected. The plume resembled a mist of trace chemicals largely invisible to the eye, rather than a river of oil.
In normal circumstances, crude oil floats to the surface, where it can be skimmed, burned off or evaporated. Floating on the waves, it can be churned into smaller drops readily digested by bacteria.
But oil from the ruptured well, broken down by sprays of chemical dispersants and held at depth by water pressure, has formed microscopic droplets not buoyant enough to break through the transition layer that separates warm surface currents from the cold bottom water, several experts said.
Dr. Valentine found none of the main oil-eating microbes in the deepwater plumes he tested. Whether that is because the Corexit killed them, its too cold in the deep ocean, they don't like the heavier portions of the oil that don't float to the surface, or for some other reason, the result is a failure.
Scientists have already said that Corexit is dangerous to sealife and humans (see this and this), and called the application of Corexit the largest science experiment ever conducted. For example, Rob Kendall, director of Texas Tech’s Institute of Environmental & Human Health, says:
This is a catastrophe of enormous proportions. To me, this is the biggest environmental toxicology experiment we’ve ever conducted.
And Kim Withers, a coastal ecologist at Texas A&M University in Corpus Christi notes:
It's like the biggest science experiment ever. Unfortunately, it's a completely uncontrolled experiment.
They were talking about the toxic effects of Corexit on sealife.
But Dr. Valentine's findings lead me to conclude that the experiment on the potential of Corexit to help the microbes break down the oil has failed as well.
3 comments:
→ Thank you for contributing to the conversation by commenting. We try to read all of the comments (but don't always have the time).
→ If you write a long comment, please use paragraph breaks. Otherwise, no one will read it. Many people still won't read it, so shorter is usually better (but it's your choice).
→ The following types of comments will be deleted if we happen to see them:
-- Comments that criticize any class of people as a whole, especially when based on an attribute they don't have control over
-- Comments that explicitly call for violence
→ Because we do not read all of the comments, I am not responsible for any unlawful or distasteful comments.











It's astonishing to imagine how much oil must be in the Gulf, when considering the images of so much oil in the marshes, and the mats of sludgy oil and tar balls on the beaches. And there is continued arrival of more oil onshore, mixed with Corexit, although this may be new oil from the seepage, and that has been sprayed from the air in recent weeks. So clearly much oil rises to the surface, and continues to rise.
ReplyDeleteI'm curious as to the C? length of the alkanes at which the buoyancy difference occurs, with respect to what rises to the surface and what remains at depth. And also the C? cut-off between meat and vegetables for Alcanivorax borkumensis.
Earlier on there had been discussion of whether the methane (and perhaps also ethane and propane?) would rise to the surface, to enter the atmosphere, or whether the water's cold temperature caused methane hydrate formation. If so, then are there plumes of methane hydrate at the bottom of the Gulf, or does it slowly rise and [dissolve/sublimate/dissociate?] back into methane, entering the atmosphere over a more extended time? What about the methane-eating organisms? I believe Samantha Joye had measured a near-hypoxic level in the plume she first discovered. Would this not lead to shutting down of bacterial activity, the methane-eating bacteria quickly using up dissolved oxygen (which is presumably low at depth)? With the methane fraction estimated at 40% of the spew, the prospect of so much of it entering the atmosphere in a short time is daunting. What effect might Corexit have on the methane? Perhaps Corexit is keeping the methane in the water.
Shouldn't Dr. Valentine also investigate the disposition of the methane? Is that included in his study? Is anyone studying the methane?
Shame on you EPA, NOAA and our Gov't in general for disseminating false information and allowing a Corporation to control/cover up all aspects of a "National Emergency" that is destroying and may ultimately end up destroying a good portion of all life in our seas/oceans and who knows what on land - including people.
ReplyDeleteShame on you.
Well here is the story about the skimmers. http://www.wlox.com/Global/story.asp?S=12921402
ReplyDelete"One of those tasks includes an attachment that sprays microbes to eat oil. Each vessel can spray 15,000 gallons and stay on the water for about six hours."
Thanks for posting this story.